A large number of ACVR1/ALK2 inhibitors were previously synthesised by Paul Brenner’s team (Target Discovery Center, University of Oxford) for the purpose of treating Fibrodysplasia Ossificans Progressiva (FOP). Although these compounds were not designed with blood-brain-barrier permeability in mind, they can serve as good bench-marks for my cellular assays. Therefore, 30 of these legacy compounds were chosen to be tested before other new bespoke Diffused Intrinsic Pontine Glioma (DIPG) compounds from Ontario Institute for Cancer Research (OICR) and Charles River Laboratories (CRL). Inhibition of TGFBR1/ALK5 leads to cardiac toxicity. Since I have not yet been able to establish a robust nanoBRET target engagement assay for TGFBR1/ALK5, I will have to resort to dual luciferase promoter assay (orthologous assay) for the time being.
EGFP signal in transfected cells. HEK293 were transfected efficiently. Cells can be harvested and re-seeded into 96-well plates for ALK5 dual luciferase assay.
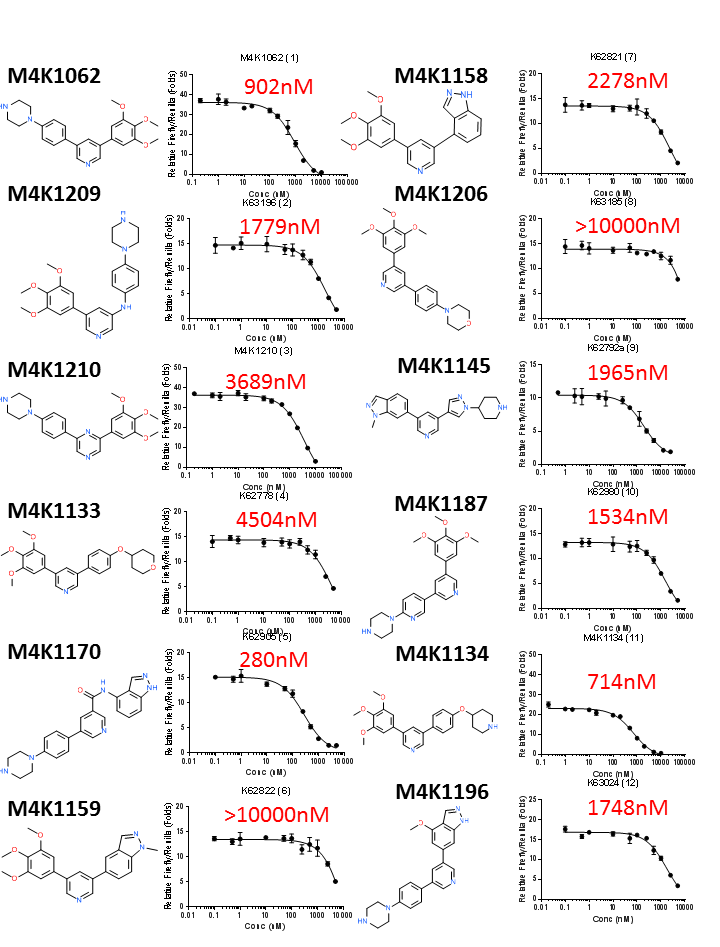
Chemical structures and DLA IC50 curves of the first 12 legacy ACVR1/ALK2 inhibitors. IC50 values estimated by GraphPad Prism are shown in red.
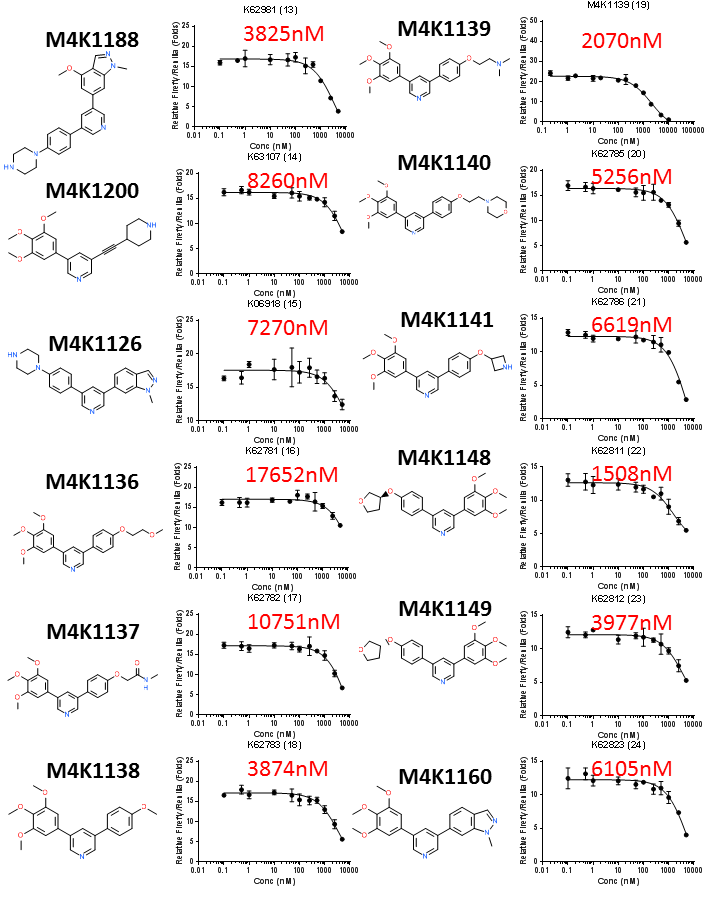
Chemical structures and DLA IC50 curves of the next 12 legacy ACVR1/ALK2 inhibitors. IC50 values estimated by GraphPad Prism are shown in red.
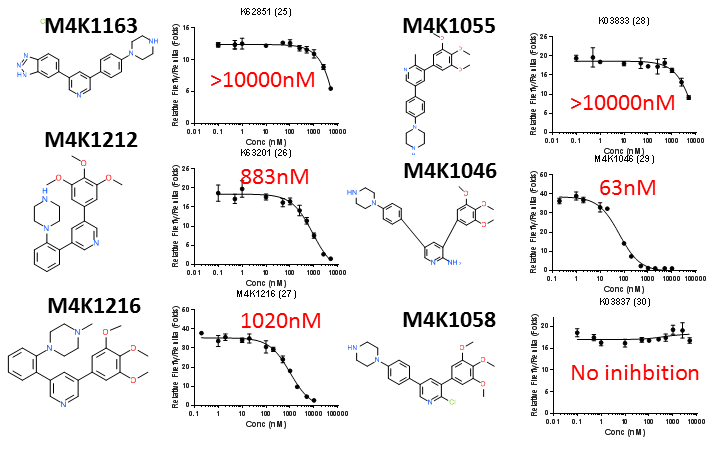
Chemical structures and DLA IC50 curves of the last 6 legacy ACVR1/ALK2 inhibitors. IC50 values estimated by GraphPad Prism are shown in red.
The off-target ALK5 inhibition by most of the legacy ACVR1/ALK2 inhibitors are not high. Only a handful of legacy compounds had ALK5 IC50 <1000nM (M4K1062, M4K1134, M4K1212 and M4K1046). Among them M4K1046 has an ALK5 IC50 of 63nM. This shows that the legacy compounds designed for FOP treatment had been well designed to minimise the risk of cardiac toxicity. However, in all cases, potency towards ALK2 and ALK5 appeared to be coupled. Differentially increasing the potency towards ALK2 while minimising ALK5 potency is highly desirable for future M4K designs. More careful analysis of the structure-activity-relationship (SAR) of these legacy compounds is necessary to draw useful lessons for new M4K compound design.
For experimental methods and details, please visit my Zenodo post.

You might consider combining two ideas/approaches with any promising compounds you might have by using magnetic nano-particles for drug delivery & ultrasound. Sources: Nanject @ https://borgenproject.org/what-is-the-nanject and “Buzzing Cancer Drug Malignancies in the Brain” @ http://rh.gatech.edu/news/611058/buzzing-cancer-drugs-malignancies-brain . Magnetic nano-particles will cross the blood/brain barrier and ultrasound opens the tumor target site for increased drug delivery. Years ago, CalTech & Children’s Hospital used magnetic nano-particles (encapsulated in a soft capsule to swallow like Contact(tm)) in an experimental approach for the treatment of Neuroblastoma(NB) by coating the magnetic nano-particles so that one-antigen/one antibody of Neuroblastoma would combine together and this was then filtered out of blood during blood transfusions using magnets. Enjoy 🙂
Thank you so much Shaw! That is definitely a possible venue that we can consider if BBB-penetrance proves to be a bottleneck.