Background
Malignant-Brain-Tumor (MBT) protein L3MBTL1 contains three tandem MBT repeats (3xMBT), which interact with mono- and dimethylated lysines of histone H4, H3 and H1.4 resulting in transcriptional repression. The interaction requires conserved aspartic acid (D355) in the second MBT repeat (PMID:17540172, 18408754, 20870725, 18408754).
Assay validation
Assay measuring the interaction of L3MBTL1 to histone H3 was developed using NanoBRET technology – a proximity-based assay that can detect protein interactions by measuring energy transfer from a bioluminescent protein donor to a fluorescent protein acceptor. At the beginning we determined the optimal donor to acceptor ratio of four donor constructs and their binding deficient mutants (D355N): C- or N-terminally NanoLuc® tagged 3xMBT domain of L3MBTL1 and full L3MBTL1 and two acceptor constructs: C-terminally HaloTag®Fusion tagged histone H3 and H4. The best results were obtained with C-terminally NanoLuc® tagged 3xMBT domain of L3MBTL1 and histone H3 at 1:100 ratio, respectively (Fig.1). To validate the assay, we treated the cells with available weak antagonists UNC1215 and UNC669 (PIMID: 23292653, 21417280). Both antagonists at high concentrations decreased the NanoBRET signal in a concentration-dependent manner (Fig 2.).
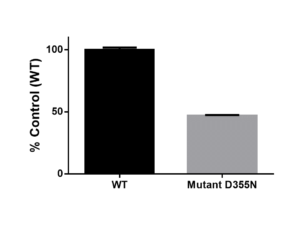
Fig.1. 3xMBT(L3MBTL1) D355N mutant shows a lower NanoBRET ratio than the wild type protein. HEK293T cells were co-transfected with C-terminally NanoLuc® tagged wild type 3xMBT(L3MBTL1) or D355N mutant and histone HaloTag®Fusion tagged histone H3- at ratio 1:100, respectively. The results are mean+/- SEM of 3 replicates.
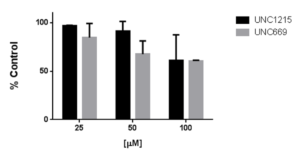
Fig.2. UNC1215 and UNC669 decrease the interaction of 3xMBT(L3MBTL1) with histone H3 in a dose-dependent manner. HEK293T cells were co-transfected with C-terminally NanoLuc® tagged wild type 3xMBT(L3MBTL1) or D355N mutant and C-terminally HaloTag®Fusion tagged histone H3. Cells were treated with indicated concentrations of antagonists for 20 h. The results are mean +/-SEM of 3 replicates.
Please go to Zenodo for experimental details.
