Background
Transcriptional coactivators p300 and CBP catalyze the acetylation of lysine residues of a large variety of histone and non-histone proteins (PMID:28426192). p300 and CBP are considered functional homologous proteins that share around 86% amino acid sequence identity in the HAT domain (PIMID:7870179). The upregulation of p300/CBP has been associated with various cancers, indicating an important role in tumorigenesis (PIMID:27881443).
Assay validation
P300/CBP has been reported previously to acetylate p53 at lysine 320 and histone H3 at lysine 27 (PIMID: 25545885, 29804834). Based on the findings, we developed p300 cellular assays measuring p53 acetylation. As shown in Fig.1 overexpression of p300 but not its catalytic mutant (Y1467F) increased p53 acetylation levels. We also developed p300/CBP cellular assay measuring H3K27 acetylation. Knocking down of p300/CBP resulted in a global decrease of H3K27 acetylation levels (Fig 2A). The H3K27 assay was further validated with p300/CBP selective inhibitor A-485 (PIMID: 29769713). A-485 decreased H3K27 acetylation levels in a dose-dependent manner in HFF cells (Fig.2B). Similar results were obtained in other cell lines (data not shown). The z factor for the assay equals 0.7.
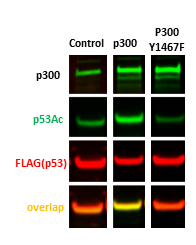
Fig.1. The wild type but not catalytic mutant of p300 acetylated exogenous p53. HEK293T cells were co-transfected with FLAG-tagged p53 and empty vector(control), p300 wild type, or p300 Y1467F catalytic mutant for 24 h. The acetylation levels were analyzed in Western blot. (Taylor Mitchell)

Fig.2. The loss of p300/CBP activity results in a decrease of H3K27 acetylation levels. A. p300/CBP knock-down. HEK293T and MCF7 cells were treated with siRNA targeted for p300/CBP and control siRNA for 3 days. B. p300/CBP inhibition. HFF cells were treated with p300/CBP selective inhibitor A-481 at indicated concentrations for 3 days. H3K27 acetylation levels were determined by Western blot. (Dalia Barsyte, Magdalena Szewczyk)
Experimental details are posted on Zenodo.
