In the second purification round of N-HEAT_81-1643, I added an additional purification step with heparin resin. My aim with doing this additional step was to improve the purity of the HTT N-HEAT_81-1643 sample from nucleic acid material/other proteins. Similar experiments proved successful for full HTT+HAP40 sample (by Dr. Rachel Harding). Our results showed that the additional step with heparin does not change the purity, oligomeric state, or the content of nucleic acid in the sample of N-HEAT_81-1643 (A260/280 = 0.7).
I also performed DSF, DSLS and DLS experiments with HTT N-HEAT_81-1643. The DSLS experiments show the construct aggregates at high temperatures around 80 °C. The DSF experiments showed the construct has a melting temperature around 46 °C. No significant difference in the melting temperature (Tm) or temperature of aggregation (Tagg) was observed in the two buffer systems or protein concentrations tested.
The DLS experiments show that the HTT N-HEAT_81-1643 particles in solution have a radius of around 15 nm, implying the presence of large particles. These results are consistent with gel filtration chromatography results, which show N-HEAT_81-1643 elutes as a very large particle in solution (early elution volume). However, there is still the possibility that the construct HTT N-HEAT_81-1643 is in an extended conformation or in complex with nucleic acid material, which would also result in large particle size readings by DLS and gel filtration chromatography. So, in future experiments we will run negative stain experiments, which will tell us more about the conformation/oligomeric state of the sample.
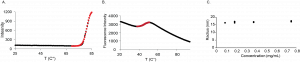
Biophysical characterization of HTT N-HEAT_81-1643 by DSLS, DSF and DLS. Representative curve of the DSLS experiments with HTT N-HEAT_81-1643 (0.2 mg/mL) in Buffer 1 (A) and the DSF experiments with HTT N-HEAT_81-1643 (0.1 mg/mL) in Buffer 2 (B). C. DLS results for HTT N-HEAT_81-1643 tested at different concentrations (n = 2).
