Recently, a study of cancer-focused protein-protein interactions identified a direct interaction between NSD3 and the oncoprotein Myc in the non-small cell lung cancer cell line H1299 (Li, Z et al. 2017). Importantly, the study found that increased NSD3 expression levels can activate Myc in H1299 cells as measured by an E-box luciferase reporter. However, the functional significance of this activity in the context of lung cancer remains unclear.
To study NSD3 in this context, as well as other cell types, I have been optimizing lentiviral vectors to express a tagged NSD3short transgene. Initially, I had limited success producing viral particles capable of integrating a bicistronic expression cassette containing NSD3short and a puromycin resistance marker separated by an internal ribosome entry site (IRES). I hypothesized that this may be due to packaging constraints of the lentiviral system and opted to decrease the size of my construct by replacing the IRES element with a sequence encoding a T2A self-cleaving peptide (details of mutagenesis can be found here – Zenodo – Exp016). The 2A self-cleaving system has the added benefit of equimolar expression, while the IRES sequence tends to favour expression of the first coding sequence at approximately 3:1 (Goedhart et al. 2011). For a helpful overview of this topic, I suggest checking out an excellent blog post on the subject over at addgene. This modification to the initial construct improved production of functional virus, as I was able to select for stable integration and expression of NSD3short in multiple cells lines (full experimental details can be found here – Zenodo – Exp018). Interestingly, overexpression of either NSD3short or a PWWP1 mutant (W284A) deficient in H3K36me2 binding resulted in altered morphological features in H1299 cells (Figure1). In cells overexpressing NSD3short, I observed greater eccentricities in their appearance with some cells displaying long projections. This may be indicative of an epithelial-mesenchymal transition (EMT), which would be in line with elevated Myc activity (Cho, KB et al. 2010). EMT is a developmental switch that can promote invasion and migration of cancer cells, phenotypes associated with metastasis. However, before we can make any claim with respect to NSD3’s involvement in this process, I will have to follow up these observations by assaying hallmarks of EMT, like downregulation of E-cadherin, with multiple biological replicates.
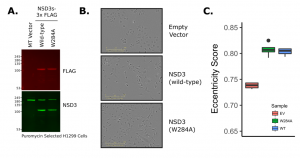
Figure 1. Expression of NSD3short-3xFLAG in H1299 Lung Cancer Cells (A) Western blot of NSD3 and FLAG in stable cell lines (B) Representative phase contrast images (C) Boxplot of cellular
eccentricity (mean of 4 images across 4 wells per sample).

This looks exciting! Is EMT the top candidate process reflected by the morphological change that you are observing, or is it one of many equiprobable interpretations?
I’m sure there are other very sensible explanations for the phenotype. Also, important to keep in mind that EMT occurs along a spectrum, the degree to which NSD3 may (or may not!) influence this process and what that means in the context of a cancer cell are big questions that will require a lot more work to sort out. Early days, but I think given what we do know about NSD3 & Myc signalling, it’s an interesting hypothesis to pursue.